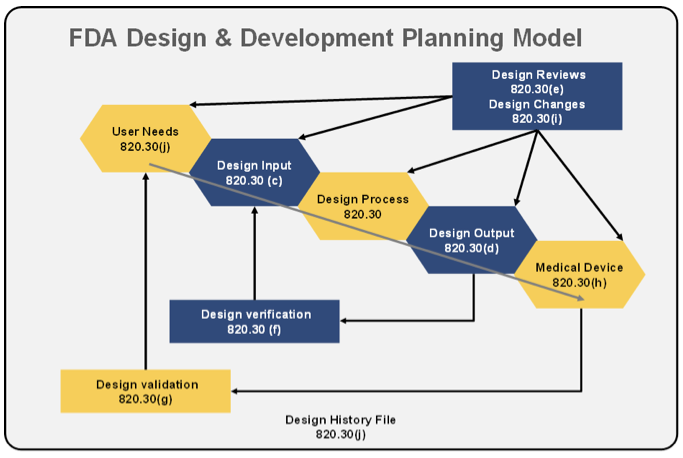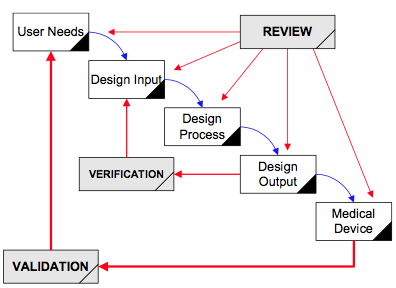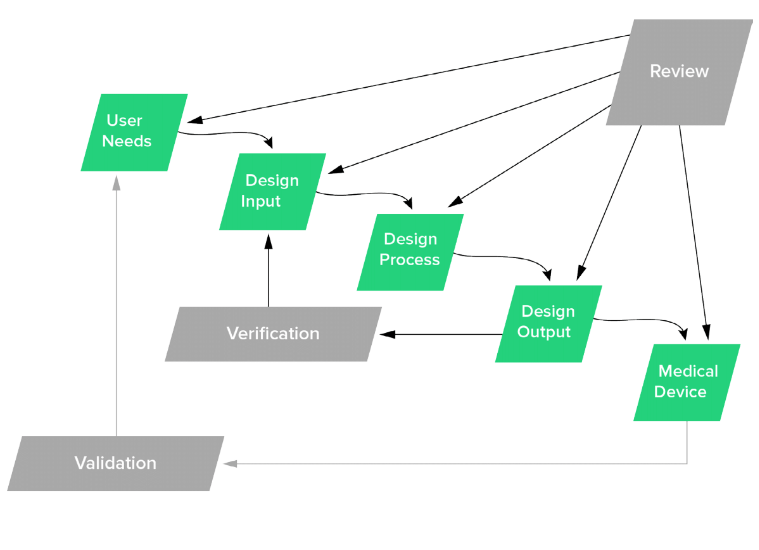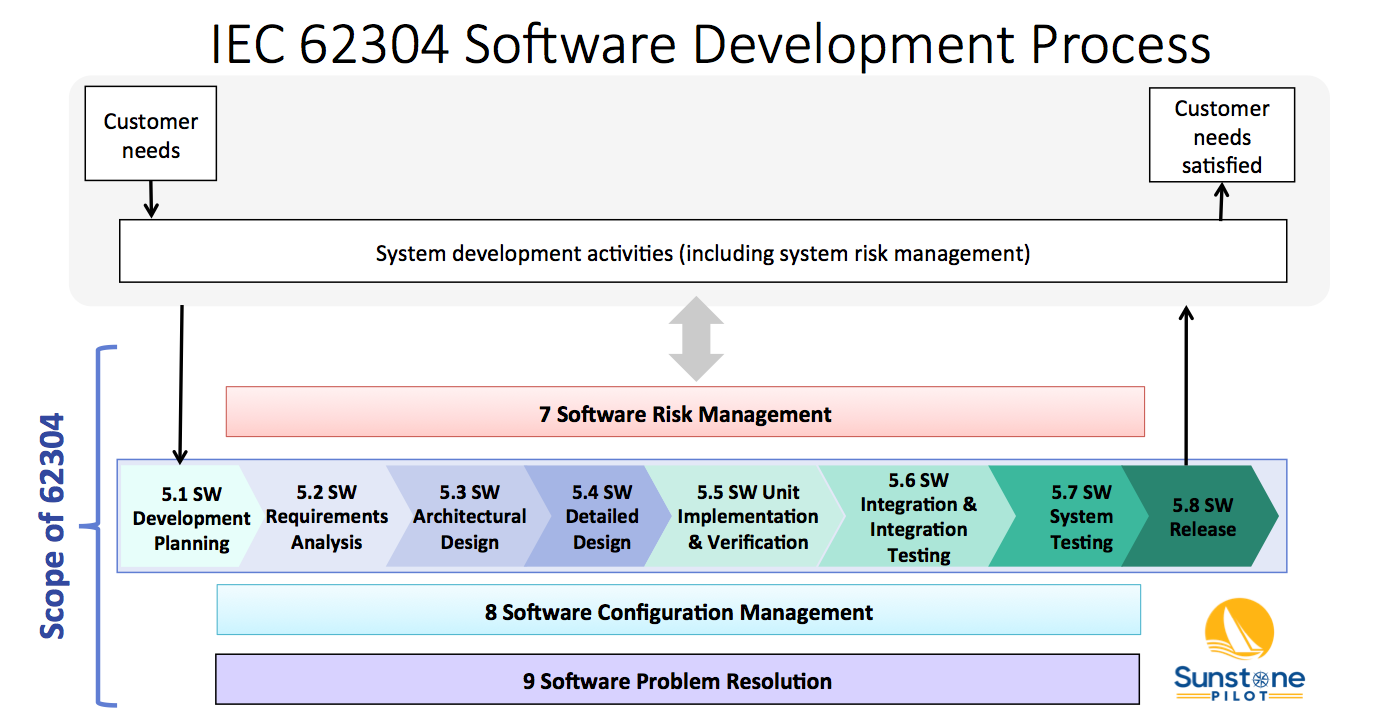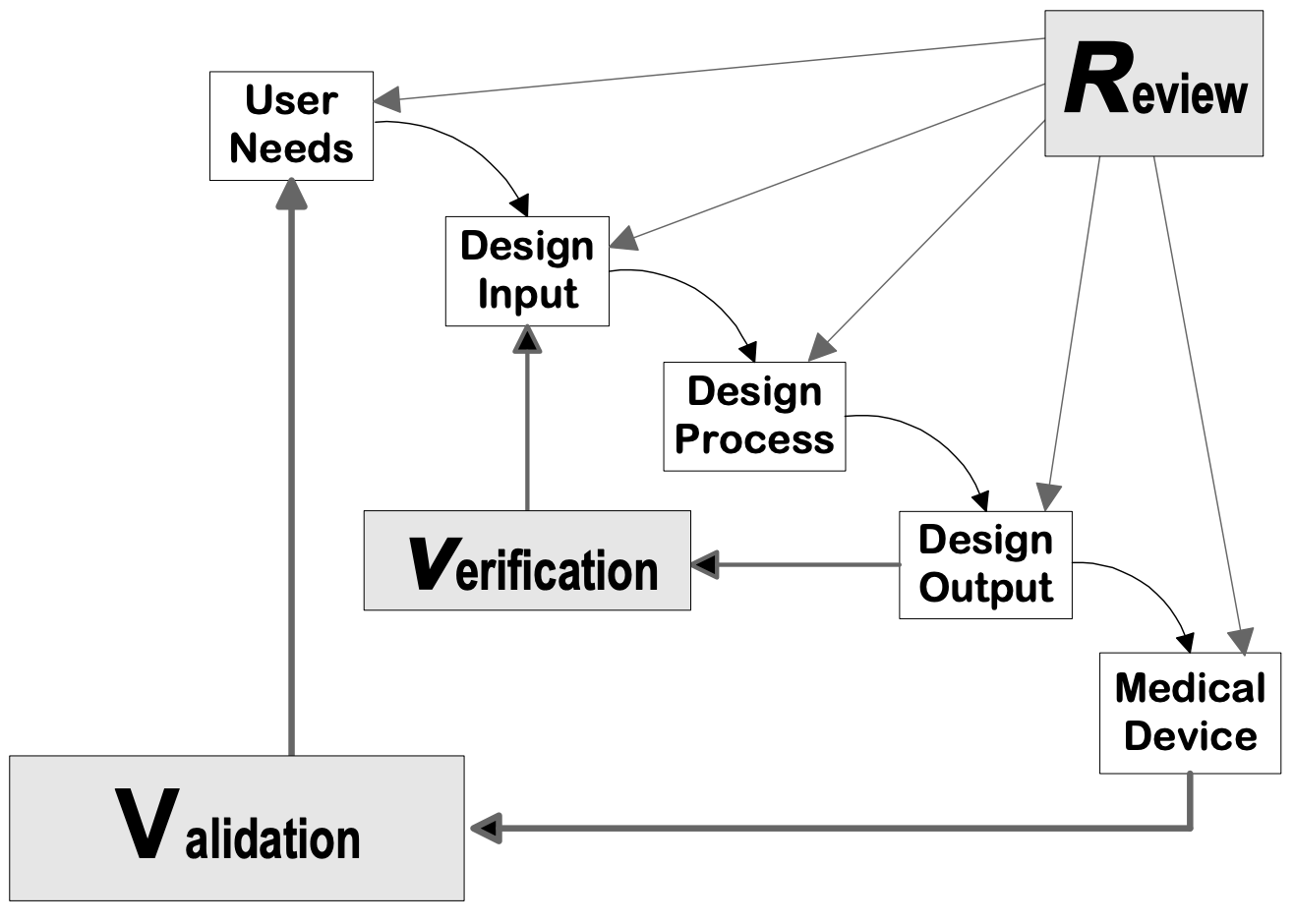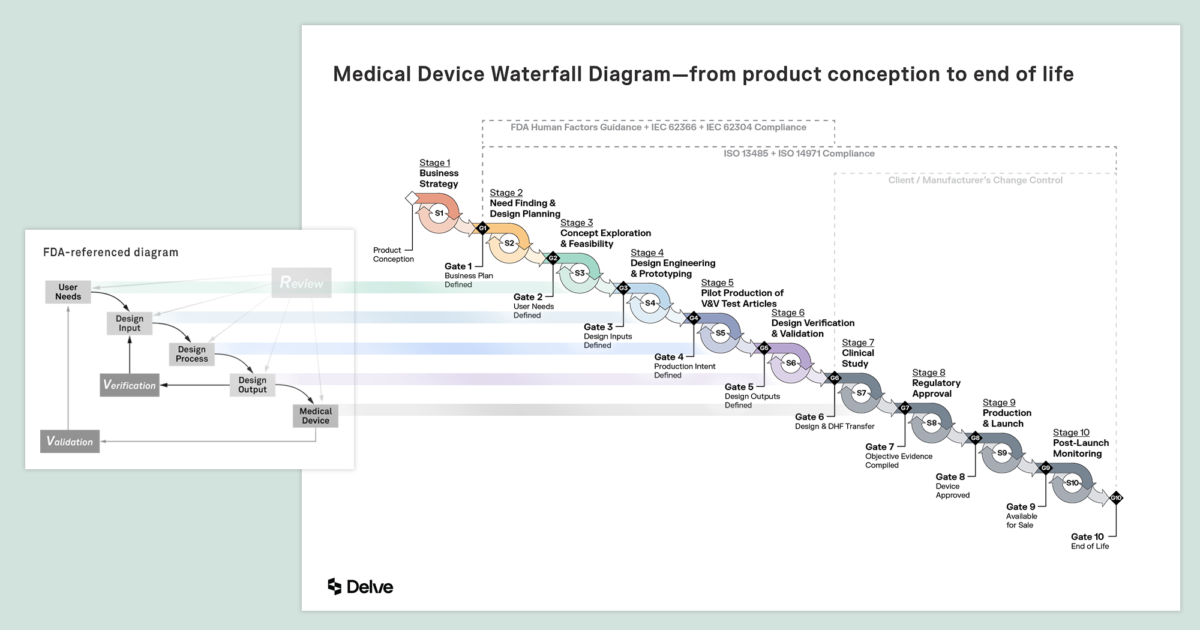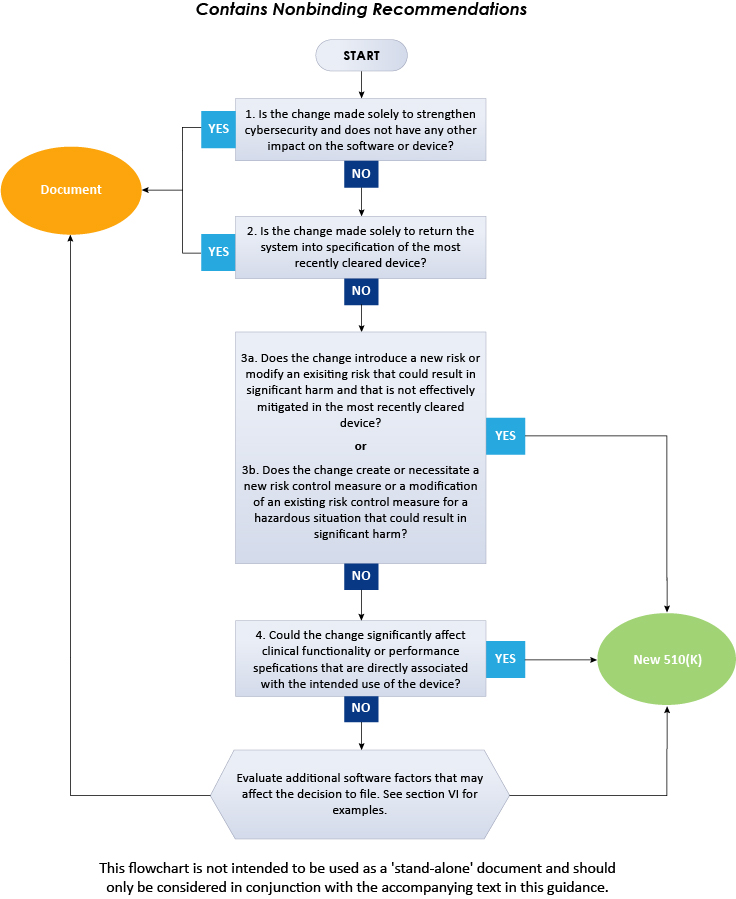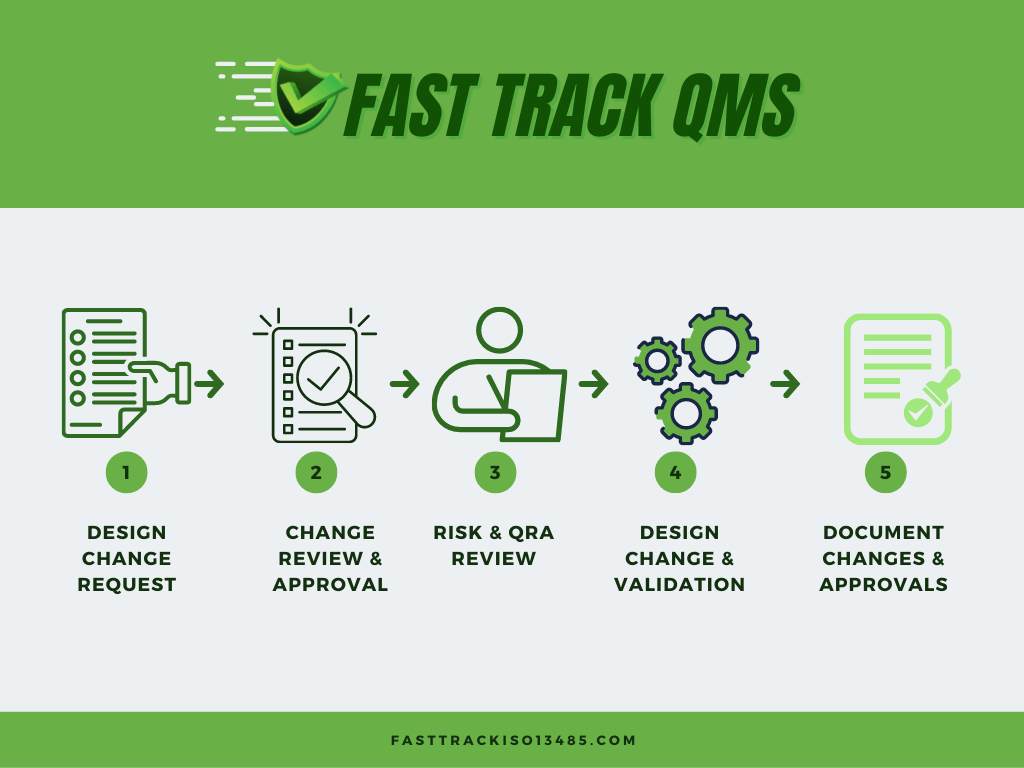
Fast Track ISO 13485 | How to Control Design Changes for your Medical Device and meet ISO 13485 requirements

ICH Q12 Examples – Pharmaceutical Product Lifecycle Management Examples – FDA Guidance – Quality by Design for Biotech, Pharmaceutical and Medical Devices

FDA Validation requirements for medical devices | Risk management, Change control, Statistical process control

AI developers should build robust change control protocols despite absence of FDA guidance | BioWorld

.webp)